APICA (also known as SDB-001, and 2NE1) is a novel synthetic cannabinoid that produces modified cannabis-like effects when administered. It has been shown to act as a potent agonist for the CB1 and CB2 cannabinoid receptors.The name “2NE1” appears to be a reference to the South Korean all-girl K-Pop group, a naming convention shared by the closely related chemical AKB48. In 2011, the two chemicals were first identified in Japan as a mixture in a product sold under the name “Fragrance Powder”. APICA has since been available for sale as a grey-area research chemical through online vendors. Synthetic cannabinoids are commonly smoked or vaporized to achieve a quick onset of effects and rapid offset. There is little information available about the use of APICA via other routes of administration, although as with other synthetic cannabinoids it could be expected to be orally active when dissolved in a lipid, which may significantly extend its duration. It is insoluble in water, but dissolves in ethanol and lipids.[citation needed] Unlike cannabis, the chronic abuse of synthetic cannabinoids has been associated with multiple deaths and more dangerous side effects and toxicity in general. Therefore, it is strongly discouraged to take this substance for extended periods of time or in excessive doses. Thorough independent research and harm reduction practices are strongly advised if choosing to use this substance.
Chemistry
APICA, or N-(1-adamantyl)-1-pentyl-1H-indole-3-carboxamide, is a synthetic indole cannabinoid. Like many synthetic cannabinoids, it can be considered to be composed of four linked structures: core, bridge, head, and tail. In APICA, the core indole group is substituted at R1 with a pentyl chain tail and at R3 with a carboxamide bridge linking to an adamantyl head. APICA can be considered an analog of both AKB48, which features an indazole in place of APICA’s indole group, and STS-135, in which the pentyl tail is further substituted with a terminal fluorine.
Pharmacology
APICA acts as a full agonist of the cannabinoid receptors, with similar potency at both CB1 and CB2. In vivo experiments measuring the response of rats to APICA and similar drugs found that APICA had a similar potency to Δ9-THC and around a third the potency of JWH-018. In comparison to these other cannabinoids, APICA appeared to elicit a longer duration of effect. Caution should be exercised in interpreting studies using animals, however, as effects may differ significantly in humans.[5Pharmacological studies have reported EC50 values of 6.89 ± 0.11nM at CB1 and 7.54 ± 0.11nM at CB2. It is likely that APICA shares many of the in vivo properties of Δ9-THC. However, the role of these interactions and how they result in the cannabinoid high has yet to be scientifically validated. In vivo metabolic studies on APICA have shown that the drug is fully metabolized with none of the original compound detectable in urine. The major metabolites were mono- or dihydroxylated on the adamantyl ring system and monohydroxylated on the pentyl chain.
Legal Status
As of October 2015 APICA is a controlled substance in China.



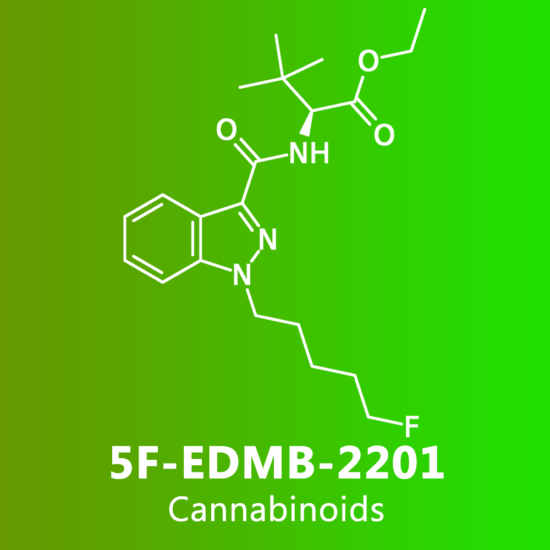
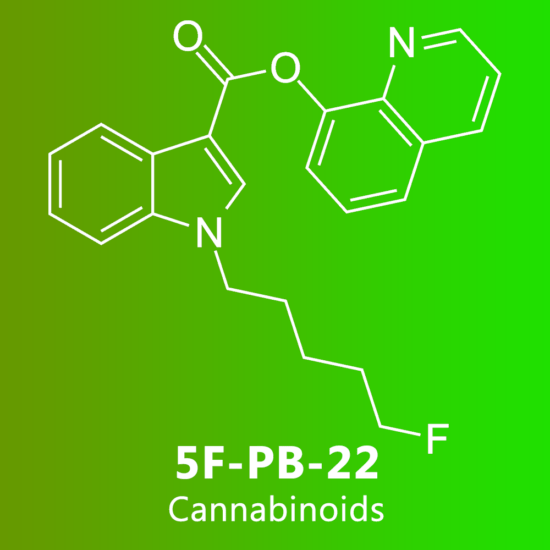
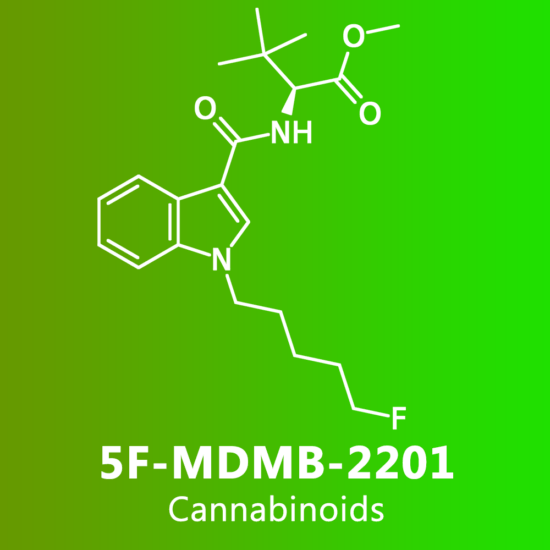
Reviews
There are no reviews yet.