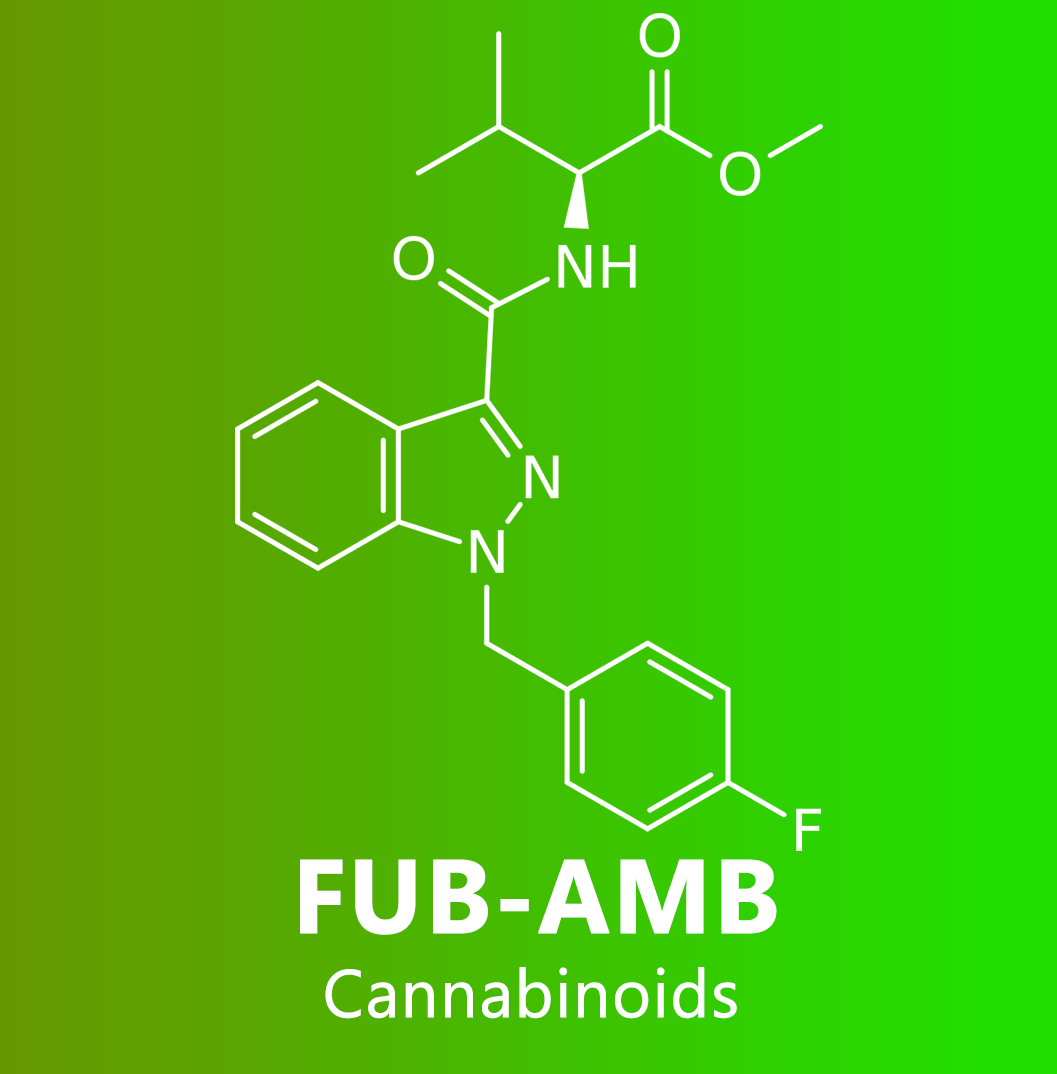
AMB-FUBINACA (also known as FUB-AMB and MMB-FUBINACA) is an indazole-based synthetic cannabinoid that is a potent agonist for the cannabinoid receptors, with Ki values of 10.04 nM at CB1 and 0.786 nM at CB2 and EC50 values of 0.5433 nM at CB1 and 0.1278 nM at CB2 and has been sold online as a designer drug. It was originally developed by Pfizer which described the compound in a patent in 2009, but was later abandoned and never tested on humans. AMB-FUBINACA was the most common synthetic cannabinoid identified in drug seizures by the Drug Enforcement Administration in 2017 and the first half of 2018.
Legal status
n the United States, AMB-FUBINACA is a Schedule I Controlled Substance. Prior to being scheduled at the federal level, the state of Louisiana banned AMB-FUBINACA through an emergency rule after it was detected in a synthetic cannabis product called “Train Wreck 2” which had been linked to adverse events and seizures on 3 June 2014.
Sweden’s public health agency suggested classifying AMB-FUBINACA as a hazardous substance on November 10, 2014.

Reviews
There are no reviews yet.